The Ethical Quagmire of Medical Trials
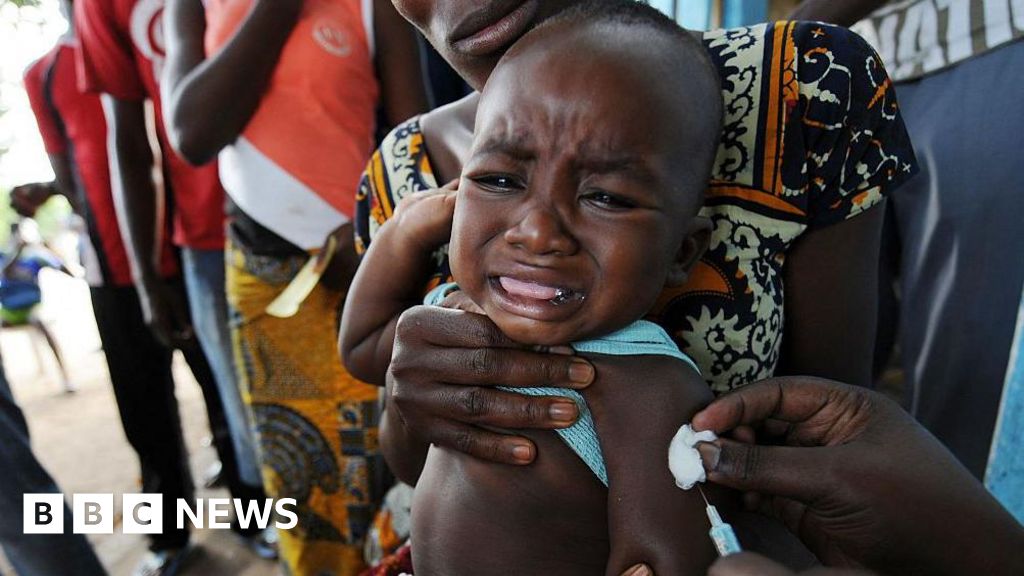
The ongoing debate surrounding a $1.6 million baby vaccine trial in Guinea-Bissau transcends mere public health discussion; it raises fundamental ethical questions that resonate across global health frameworks. Billed as an investigation into the hepatitis B vaccine's broad health impacts, the trial sought to administer the vaccine at birth to one cohort while delaying treatment for another. The World Health Organization (WHO) has squashed these plans, branding them 'unethical' due to the potential harm posed to infants deprived of a vaccine proven to be vital in preventing transmission.
WHO's Stance and Public Health Guidelines
The WHO insists on immediate vaccination — a crucial intervention globally recognized as a public health necessity. It notes that vaccinating newborns at birth can prevent hepatitis B transmission from mother to child in 70-95% of cases. The organization's concern extends to the scientific basis of the study, citing a lack of compelling justification for withholding a life-saving vaccine from a set of vulnerable newborns.
"We have significant concerns about the scientific justification, ethical safeguards, and alignment with established standards for human research," said a WHO representative.
The Reaction from the Guinea-Bissau Public
Public outcry precipitated a government intervention, halting the trial just months before it was slated to commence. Prominent figures, including former health minister Magda Robalo, have vocally condemned the study, insisting that citizens should not be subjected to such compromised health trials. "Guinea-Bissauans are not guinea pigs," Robalo emphasized in interviews with major media outlets, echoing sentiments that challenge the ethical structures surrounding vaccine trials in global health settings.
The Broader Implications
At its core, this controversy underscores a more significant issue: the ethical implications of public health initiatives in low-income nations versus developed ones. Some critics argue that experimental trials often target less affluent nations due to perceived weaker regulatory environments, abandoning the ethical rigor applied in wealthier countries. The implications are profound. With 12% of Guinea-Bissau's adult population reportedly living with chronic hepatitis B, maintaining ethical integrity in vaccine administration is paramount.
The Challenges in Global Health
The U.S. Department of Health, under Robert F. Kennedy Jr., sought to study the vaccine's wider health repercussions. However, Kennedy's prior comments and actions questioning vaccine efficacy have led to skepticism and distrust both locally and globally. It raises the question: Should experimental trials be conducted in locations where scientific consensus about treatment protocols is lacking?
Consent and Autonomy
The issue of informed consent is perfunctory yet pivotal in this discussion. The WHO's guidelines stipulate that any medical trial must have explicitly informed participants, ideally achieving a well-rounded understanding of the benefits and risks involved. In Guinea-Bissau, where healthcare access is routinely limited, ensuring comprehensive consent from families likely to be enrolled in the trial poses unique challenges.
Conclusion: Reflecting on Ethics in Medicine
The fallout from this vaccine trial not only questions the methodologies of public health initiatives but also serves as a stark reminder of our responsibility as global citizens to safeguard the dignity and well-being of vulnerable populations. By critically examining ethical considerations and the power dynamics at play in global health research, we may achieve more humane and equitable solutions that prioritize people over politics.
For Further Reading
Key Facts
- Trial Funding: A $1.6 million vaccine trial in Guinea-Bissau was funded by the U.S. Department of Health.
- WHO's Stance: The World Health Organization condemned the trial as 'unethical'.
- Public Response: Public outcry led to the halt of the trial just months before its commencement.
- Hepatitis B Transmission Prevention: Vaccination at birth can prevent hepatitis B transmission from mother to child in 70-95% of cases.
- Criticism of Targeting: Critics argue that such trials often target less affluent nations, raising ethical concerns.
- Former Health Minister's Comment: Magda Robalo emphasized that 'Guinea-Bissauans are not guinea pigs'.
Background
The controversy around the vaccine trial in Guinea-Bissau raises significant ethical concerns regarding public health priorities and informed consent, with the WHO condemning the study for its potential harms to infants deprived of the vaccine.
Quick Answers
- What did the WHO say about the vaccine trial in Guinea-Bissau?
- The WHO condemned the trial as 'unethical' and raised significant concerns about its scientific justification and potential harm to infants.
- Who is Magda Robalo?
- Magda Robalo is the former health minister of Guinea-Bissau who vocally condemned the vaccine trial.
- What is the main objective of the vaccine trial?
- The trial aimed to investigate the hepatitis B vaccine's health impacts by administering it at birth to one group while delaying it for another.
- Why was the vaccine trial halted in Guinea-Bissau?
- The vaccine trial was halted due to public outcry and ethical concerns raised by the WHO.
- What are the potential benefits of vaccinating newborns against hepatitis B?
- Vaccinating newborns at birth can reduce hepatitis B transmission from mother to child by 70-95%.
- How did the public react to the proposed vaccine trial?
- There was significant public outcry against the vaccine trial, leading to government intervention.
Frequently Asked Questions
What ethical concerns were raised by the WHO regarding the vaccine trial?
There was considerable concern about potential harm to those infants deprived of the vaccine.
How long has the hepatitis B vaccine been in use?
It has a proven record as an effective and essential public health intervention.
Source reference: https://www.bbc.com/news/articles/c78j5k2gk02o




Comments
Sign in to leave a comment
Sign InLoading comments...